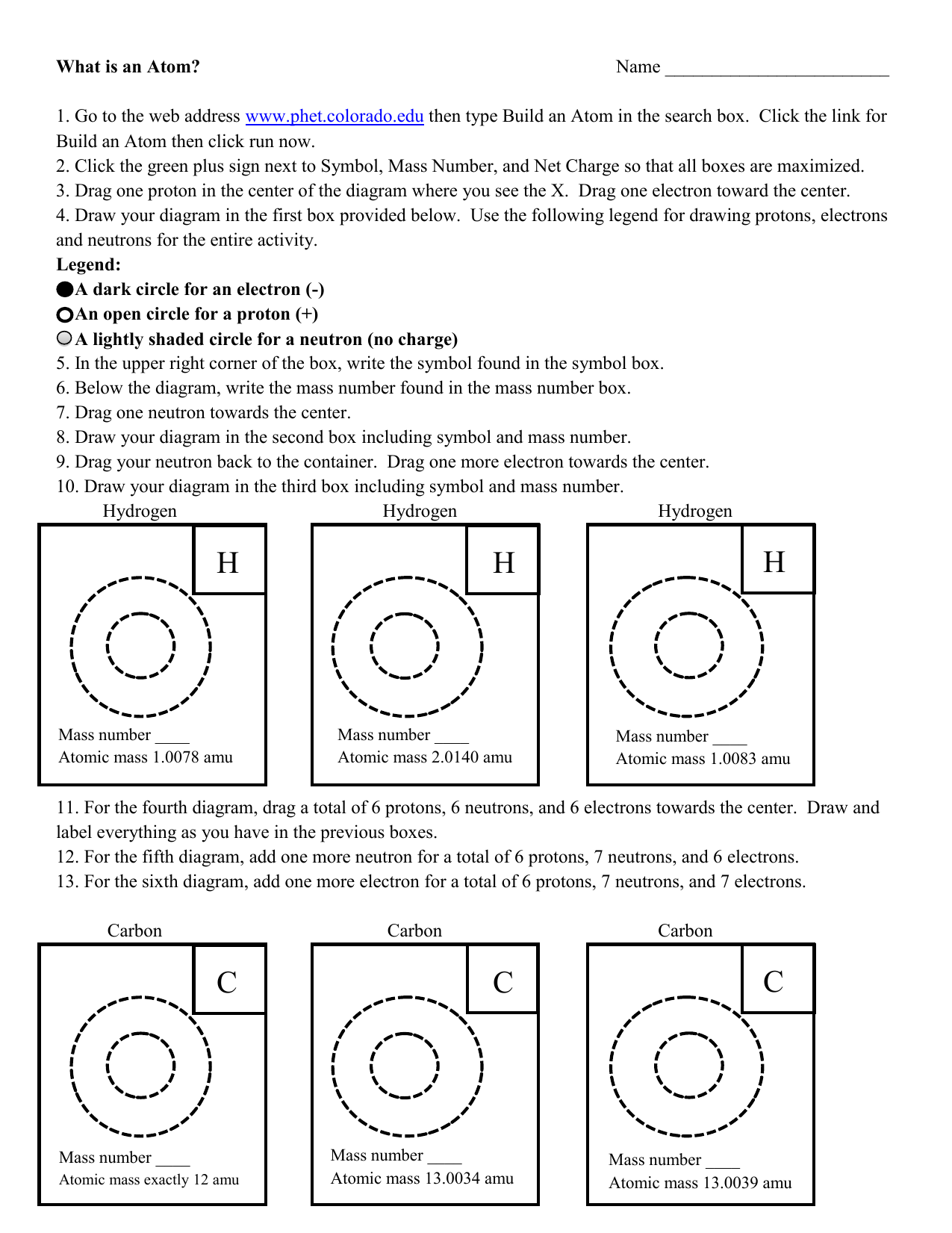
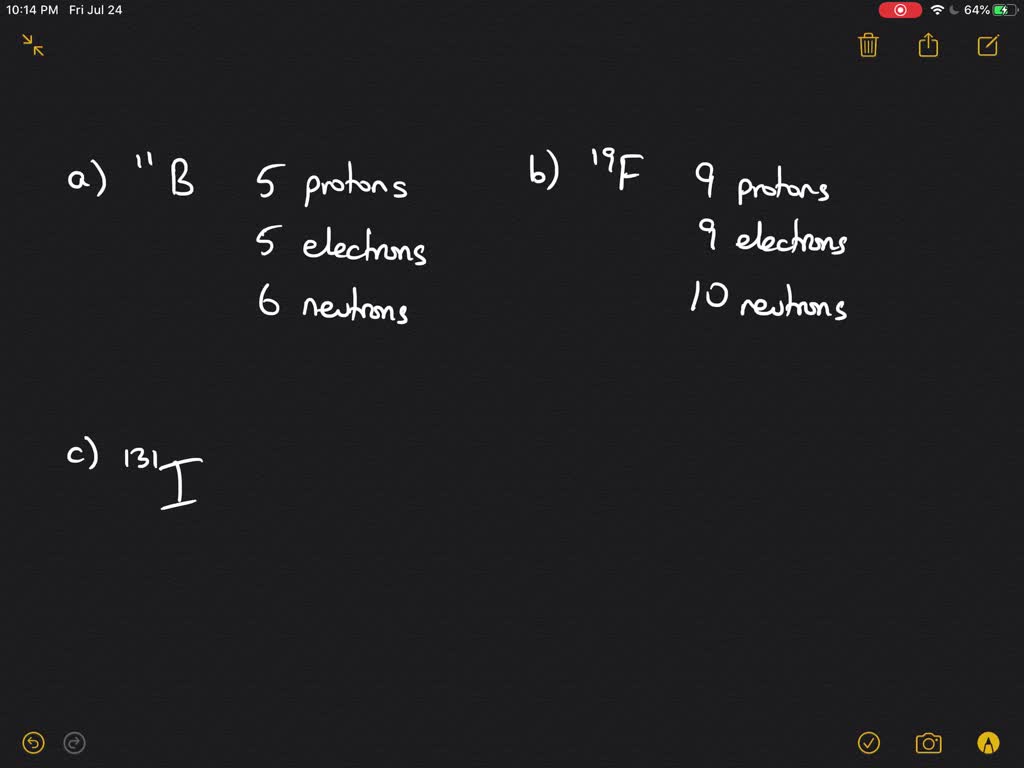How Big is an Atom? Imagine that you could increase the size of an atom to make it as big as an orange. - ppt video online download

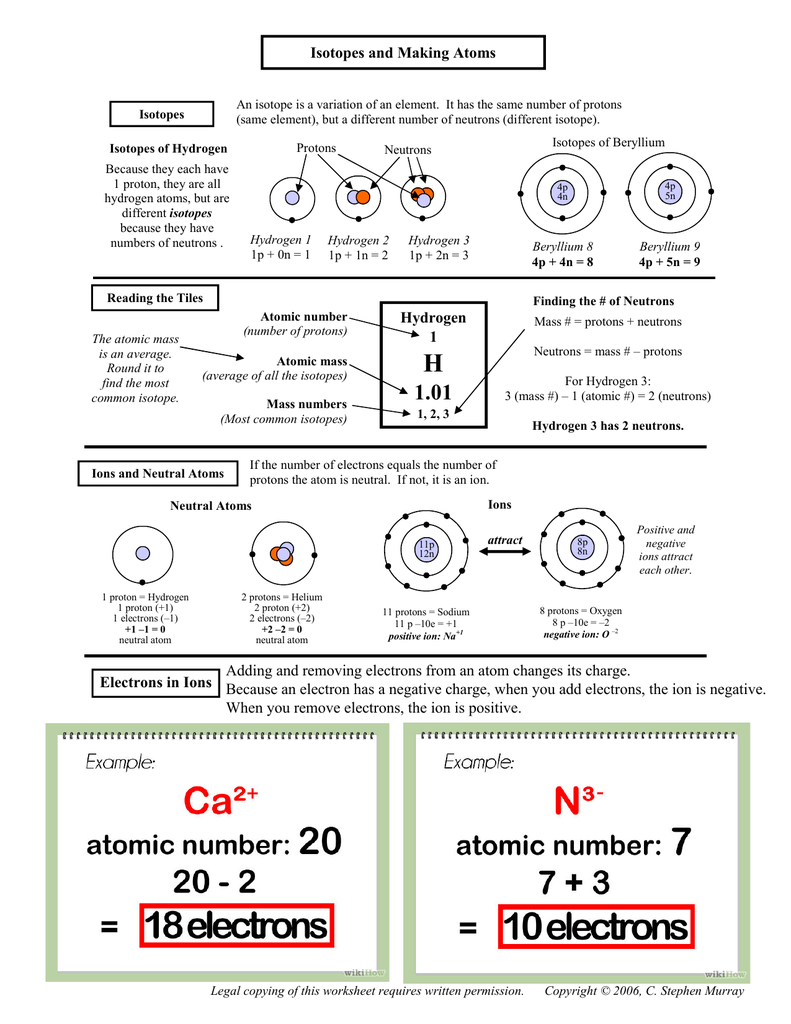
UNIT VIII Atoms and the Periodic Table Lesson#1 Atomic Number, Atomic mass, Isotopes review. - ppt download

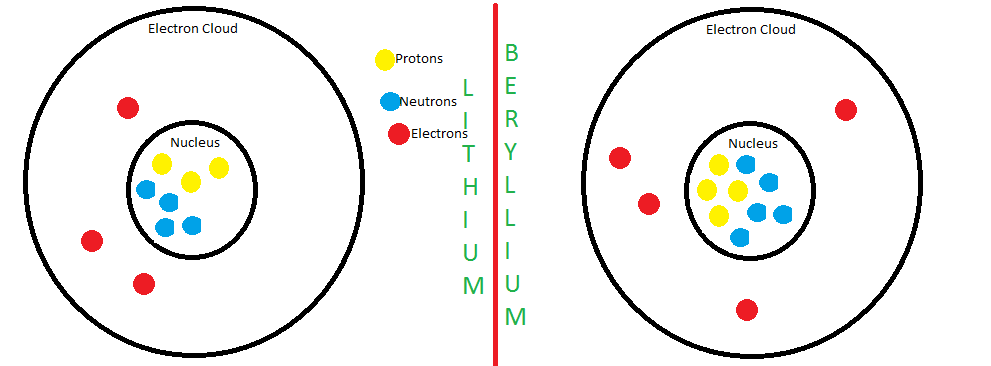
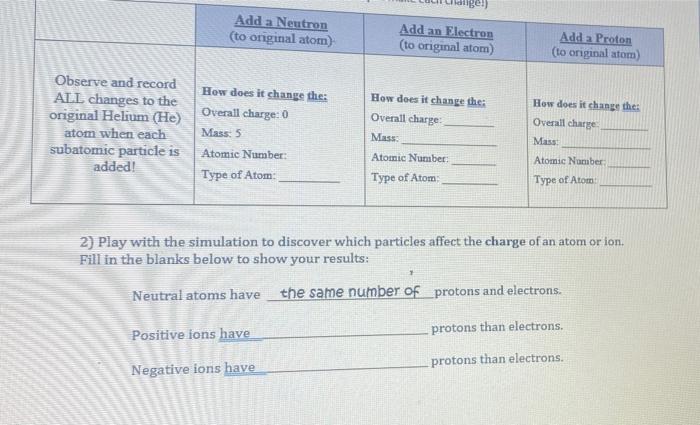
Nuclear energy Summary of period 1. Parts of the atom Mass (A.M.U)Charge Proton1+1 Neutron10 Electron1/1830 Add/subtract a proton: creates a different. - ppt download
Is it theoretically possible to add and subtract the neutrons and protons from the nucleus of an atom to change it into a different element? - Quora
Is it theoretically possible to add and subtract the neutrons and protons from the nucleus of an atom to change it into a different element? - Quora

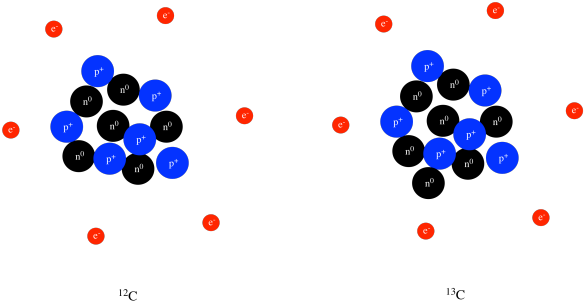
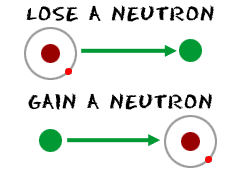
True or false? Adding one neutron to the nucleus of an atom converts it to an isotope of the same element. | Study.com